Designing to meet 3rd edition medical safety standards
One significant change in the 3rd edition standard is the additional MOOP protection
First published in 1977, the internationally accepted IEC 60601-1 standard has been continuously developed to help alleviate safety issues relating to all types of medical equipment. The 3rd edition of this medical safety standard was first published by the IEC in 2005 (IEC60601-1:2005) and was adopted by the European Union in 2006 and published as EN60601-1:2006. The USA version was also published in 2006, but unlike 2nd edition, not by UL. It is published by the American Association for Medical Instrumentation (AAMI) and appears as ANSI/AAMI ES60601-1:2006. Canada published the standard in 2008 as CAN/CSA60601:2008.
As many OEMs look to design medical devices to meet 3rd edition, many key factors need to be considered. A critical safety part of any electrical design is the AC/DC power supply. The AC/DC supply provides the necessary safety protection for the entire system to which it is connected. Understanding the regulatory guidelines of 3rd edition is critical before starting the design,
One significant change in the 3rd edition standard is the additional MOOP protection. In 3rd edition we now find that you can use Means of Operator Protection (MOOP) instead of Means of Patient Protection (MOPP). This means that the end device will not be in the vicinity of the patient and that only operators will have access to it. The majority of new AC-DC power supplies sold for medical applications are now being approved to 3rd edition with MOOP certifications.
For example, here at XP Power we made the decision to certify all of our current medical power supplies to 3rd edition (with 2 x Means Of Patient Protection on the majority of the power supplies) and also to the supplies test against the older 2nd edition. The rationale here is that following 2 x MOPP in 3rd edition is equivalent to 2nd edition in terms of separation distance, insulation schemes and dielectric strength requirements. This means that the OEM will be able to claim the safety of the power supply is at least as good as the current standard (2nd edition) and will still maintain their end equipment 2nd edition approval, even with a 3rd edition (2 x MOPP) approved PSU. (see clause 3.4 and clause 54 in UL60601-1:2003)
While the 2nd edition simply addressed basic safety issues to ensure freedom from any electrical, mechanical, radiation, and thermal hazards, it did not require devices to remain functional. Being ail-safe was adequate, and compliance with test criteria relied upon a pass/fail result that did not take into account the essential performance of the device-under-test. Recognizing these limitations, the 3rd edition introduces specifications for "essential performance" that requires equipment to continue functioning as its designers intended throughout the test process.
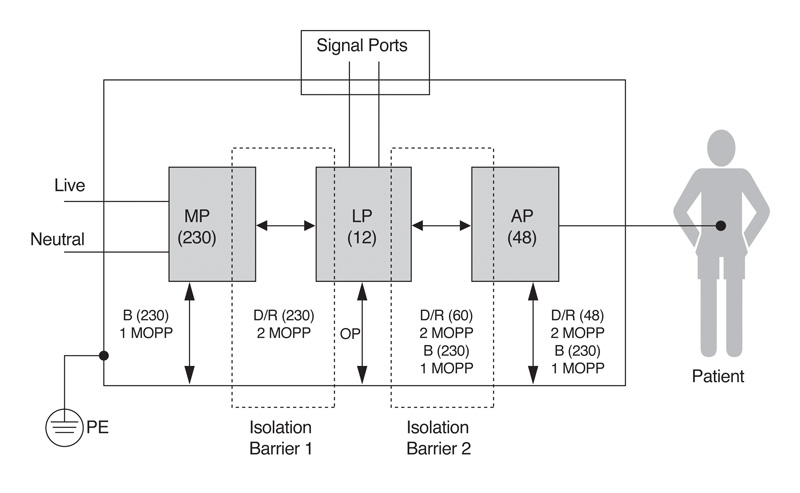
Within the electrical safety arena, the standard continues to require that medical power supply equipment implements two Means of Protection (MOP) such that if a failure occurs within one area, a second mechanism safeguards the operator and/or the patient against any electric shock hazard. Figure 1 shows the insulation diagram that applies to the main circuit blocks in a typical medical device, and shows the two isolation barriers that provide the two Means of Protection that must be present within a device that may come into contact with a patient.
Click image to enlarge
Figure 1: IEC60601-1 3rd edition demands that two means of protection (MOPs), or isolation barriers exist where patients may come into contact with equipment.
The standard allows for three defensive approaches that may be used in various combinations—safety insulation, protective earth, and protection impedance. It is critical to determine several key factors from the start of the equipment design process, including its insulation class and whether it will rely upon a protective earth connection. These considerations extend to the “applied part”, if present, that is deliberately attached to the patient. Such applied parts are separately classified by the level of electric shock protection that they provide.
Significantly for power supplies, the 3rd edition distinguishes between protecting the equipment's operator and the patient within its Means of Operator Protection (MOOP) and Means of Patient Protection (MOPP) categories. This distinction can result in quite different safety insulation and isolation requirements for circuits that operators and patients may come into contact with. Applications requiring operator protection only have to meet the clearance and creepage requirements that IEC/EN 60950 specifies for general-purpose information and technology equipment. By contrast, circuitry that is classified as patient protection must meet the far more exacting requirements that the 2nd edition of IEC 60601-1 introduced. As to who determines whether it is MOOP or MOPP is up to the manufacturer and they will need to record this in the risk management file.
Choosing a power supply with only MOOP you would have to ensure that other isolation schemes are in place between the output and the patient if the equipment is to come in contact with the patient. It complicates the design and adds cost even though the cost of the MOOP power supply might be less than an MOPP power supply. No matter whether MOOP or MOPP is chosen the standard still requires that the leakage current requirements are met. For the power supply this means 300uA in the US and 500uA for the EU. As most power engineers know if one has to modify an ITE power supply to achieve these lower leakage current levels then the emissions will be impacted and additional filtering will most likely be required to be added to the equipment. From our standpoint at XP Power, we believe that the power supply for a medical device should provide the highest degree of protection and reduce the risk of a shock hazard. Therefore we made the decision to design our power supplies to have 2 x MOPP from input the output (mains to low voltage dc). This gives the customer flexibility and assurance that they have minimized the risk of a shock hazard.
Another significant change that the 3rd edition introduces is that equipment manufacturers must now follow a formal risk management procedure that follows the ISO 14971 model, which effectively means compliance with a process standard as well as the fundamental product standard.
XP Power believes that in any medical application quality is the most critical component. To ensure our power supplies are of the highest quality, we use the industy’s most stringent derating guidelines, complete a full design verification test (DVT), use of only the highest quality components, and compile full DFMEA reports for each medical power supply that we design. We also have a full offering of BF (Body Floating) and CF (Cardiac Floating) rated supplies.