How Healthcare Demands are Impacting Power Supply Design

Medical electronics is in an era of dramatic transition. It can be easy to overlook how changes can affect technologies, such as power supplies.
Key triggers are general technological advances, an aging population, and government healthcare reforms. Trends include the proliferation of wireless technologies; an increase in implantable medical devices; a push towards home healthcare options; and greater cost-consciousness.
To meet increased density, reliability and efficiencies goals, power ratings need to be reviewed carefully. Other vital considerations include product life, thermal issues, input voltage, load, and cooling needs. Control processes for design, purchasing, and production, as well as product traceability and Corrective and Preventative Actions (CAPAs) demonstrate reliability.
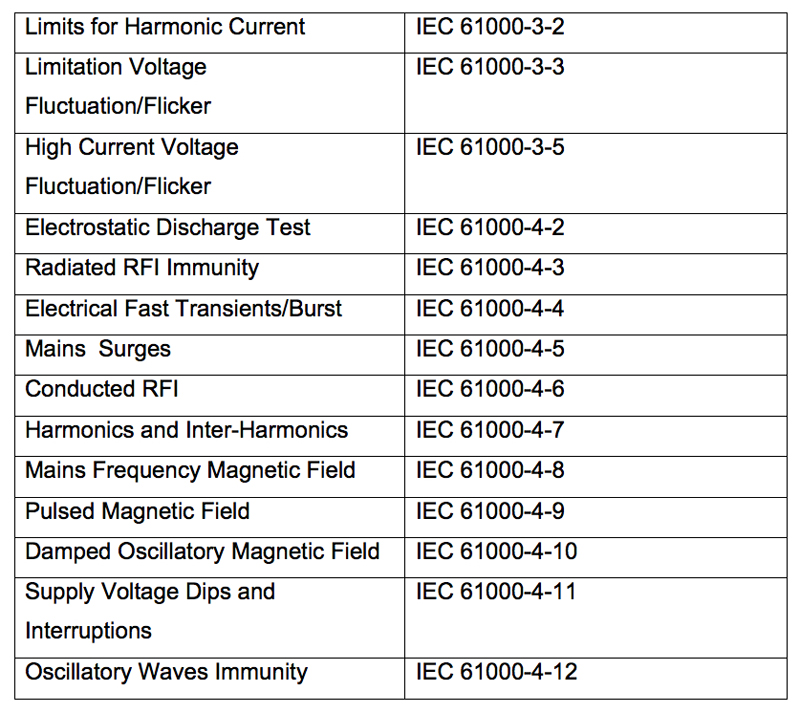
Increased regulation means designers must also consider electromagnetic compatibility (EMC) performance. EMC compliance specifications are included in various EN 61000 standards, as listed in Table 1.
Click image to enlarge
Table 1: EMC compliance standards by section
EN 61000-3-x sets limits and measures methods for low frequency emissions on the AC mains, while EN 61000-4-xx sets limits on susceptibility or immunity of the equipment powered from AC mains.
The stricter standards sustain full operation, despite a noisy, less regulated power conditioning environment in the home. For example, an infusion pump can malfunction when a mobile phone is used in close proximity, or a remote patient monitoring device may be damaged when touched by someone walking around, building up electrostatic discharge (ESD).
Regulatory Environment
Once the basic power requirements are determined, the power supply must meet standards for safety, EMC, and regulatory environmental impact compliance.
Equipment must meet stringent safety requirements and performance testing adhering to various standards, starting with IEC 60601-1 3rd Edition for medical devices. Depending on the end use, additional standards such as EN and IEC standards for EMI/EMC, RoHS compliance, and CE Mark may also apply.
When selecting an AC or DC power supply, whether off-the-shelf or custom, designers must consider the specific performance criteria for the application. It is important to bear in mind that transition to the IEC 60601-1-2 4th Edition standard on electromagnetic disturbance started in 2014 (See Figure 1).
Regulatory vs. Market Needs
Designers starting with power supplies designed from the ground-up to meet those standards will simplify the compliance process. This will also accelerate time-to-market for the end product.
Power supplies that meet the more stringent test parameters will provide a more robust power supply, potentially allowing easier system compliance to more demanding certifications (See Figure 2).
Click image to enlarge
Figure 2: Portable devices are vital for home healthcare.
The IEC 60601-1 General Standard compliance is a requirement for electrical medical equipment in many regions. The current 3rd Edition emphasises usability engineering, risk assessment, and use in home healthcare applications.
Collateral Standards
In addition to regional variations, the standard also encompasses Collateral Standards (numbered 60601-1-x) that define requirements for safety and performance. Requirements for the general standard may be over-ridden in the collateral standards. These are applicable to specific aspects of product design: namely: IEC 60601-1-2 – EMC; IEC 60601-1-3 – radiation protection for diagnostic use of X-rays; IEC 60601-1-4 – programmable electrical medical systems; IEC 60601-1-8 – alarm systems; and IEC 60601-1-11 – home healthcare environments.
Particular Standards (numbered 60601-2-x) provide supplementary definitions and requirements for specific product types, such as defibrillators, MR scanners, electroencephalograms, and insulin pumps.
Means of Protection
Medical equipment is required to incorporate Means of Patient Protection (MOPP) or Means of Operator Protection (MOOP), with patient protection being the more stringent standard. Primary distinctions relate to allowable creepage distance, isolations, and insulation. Each Means of Protection has a basic level and a 2x level, with 2 MOPP referring to the highest level of protection with 4000Vac isolation, 8mm creepage distance, and double insulation.
Increasing use of home healthcare devices blurs the lines between MOOP and MOPP. It can therefore be advantageous to design to the more stringent 2 MOPP levels, as long as the cost is not prohibitive.
Final product certification is not dependent upon whether the power supply manufacturer specifies MOOP or MOPP isolation compliance. If compliance is not specified, it will be the end product designer’s responsibility to ensure that it meets the overall requirements.
EMC Compliance Requirements
It is important for designers to review and specify a product’s performance acceptance criteria. It is not enough to just state the IEC standard. There are four levels for acceptance criteria, for normal performance within the specification limits, and degradation or loss of function, which is not recoverable due to damage of equipment or software or loss of data. In all cases, equipment shall not become dangerous or unsafe as a result of the application of the tests.
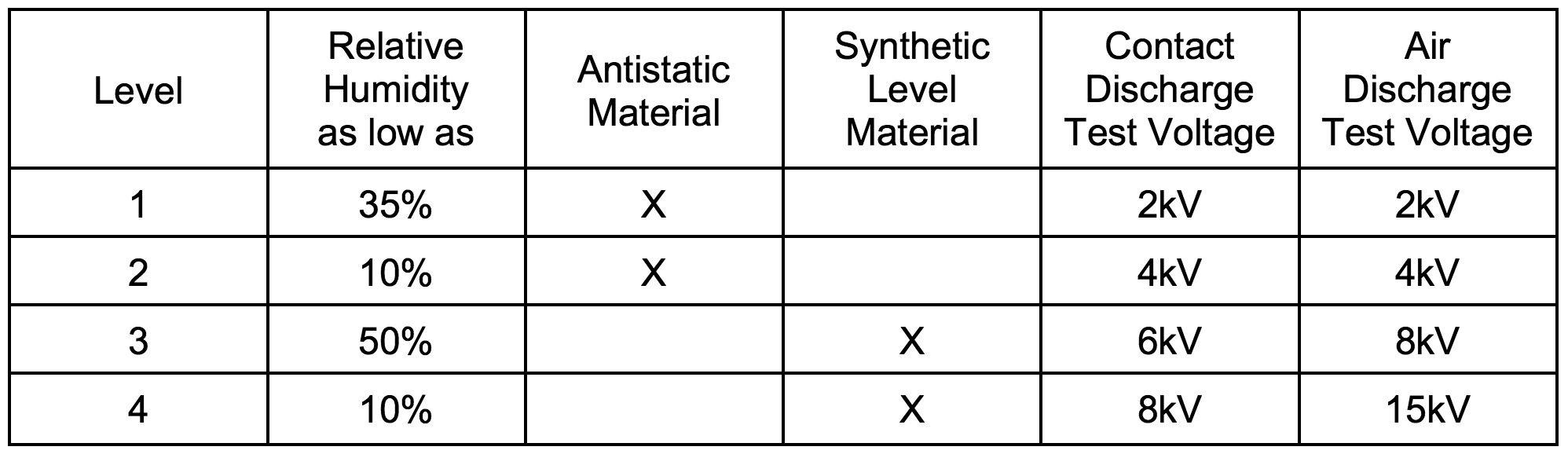
Medical device designers must pay particular attention to ESD issues. IEC 61000-4-2 standard defines four standard levels of ESD protection, using two testing methodologies (shown in Table 2).
Click image to enlarge
Table 2: EN 61000-4-2 Test Levels.
Designing a product to comply with IEC 61000-4-2 ESD requirements can be challenging at the higher discharge voltages. This becomes even more so with a Class II AC input (two wire, no-earth ground conductor). When the ESD is applied to the output or signal pin, the voltage is developed across isolating barriers and capacitors because the AC mains are virtually grounded at some point. Applied ESD voltage appears between the point where the charge is applied and earth ground.
Without careful consideration of the discharge paths within the power supply, unexpected arcing and damage can occur.
Conclusion
Market trends - RFI and wireless considerations, risk management, and regional differences - as well as issues relating to types of devices and where they will be used, present challenges. Starting with a power supply that addresses known requirements will speed development. Choosing a vendor with medical regulatory experience can further ensure the smoothest path to market.
Gresham Power Electronics