What does patient comfort have to do with the power supply?
With a global population that is both living longer and experiencing an increasing rate of chronic disease, combined with economic factors that are driving the increased of healthcare in the home, there is a growing demand for medical electrical equipment designed for use in home environment, requiring from power supplies manufacturers to not only consider standards and regulations but the patient’s comfort as well
Patient comfort and environment
Some may question, what does patient comfort have to do with the power supply?
A recent study published by the Madalena Cunha & Nélio Silva form Superior School of Health, Polytechnic Institute of Viseu, Portugal investigated noise impact on patient wellbeing in a hospital environment. The report confirmed that subjective wellbeing is influenced by the hospital noise in general and, more specifically, the noise from clinical sources (e.g. monitors, infusion pumps and other equipment).
Reducing noise in medical equipment, especially when those equipment installed in non-clinic environment becomes a must and manufacturers are now building their equipment to reduce the level of noise clause to inaudible, limiting forced ventilation to extraordinary condition.
For power supply manufacturers, that implies that power supplies will be operated with cooling limited to conduction and convection, requiring power designers to optimize performances while keeping in mind reliability in such environment. (We all know that MTBF decreases with temperature!)
Patient safety is also a very important aspect power supplies designers have to consider, and considering that studies have revealed that 60% of the homes in Europe and 40% of those in the U.S. have no reliable ground wires, it is very important to protect users of home medical devices from electrical shock. Powering home healthcare equipment safely is what standards and regulations are for, of course, but something all power designers should have in mind when developing power solutions for medical applications, especially in home healthcare.
Powering home healthcare safely: IEC 60601
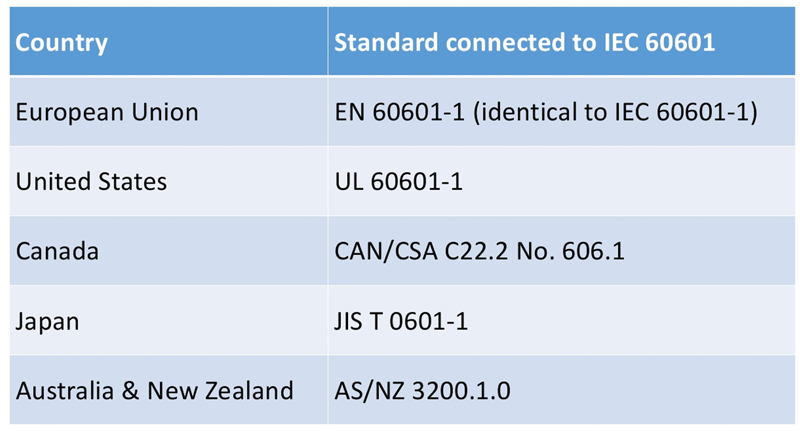
Safety is a very important concern, and medical power supplies must comply with IEC 60601 standard published in 1977. It is a standard that is internationally adopted (see Figure 1) and is continuously updated to improve patient safety and comfort. More detailed information can be found from the different standardization organizations, but it is interesting to take a quick look at the two revisions that impacted on the implementation of new technologies that power designers have to consider, guaranteeing the highest level of safety for patient and operators.
Click image to enlarge
Figure 01 – IEC 60601-1 national standards adapted by countries or regions
In 1988 the second edition introduced three categories specifying specific conditions under which the medical equipment, including the power supplies, are operated in patient vicinity (1.83 meters or 6 feet) beyond the perimeter of the intended locations of bed and treatment areas:
Type “B” (Body): No electrical contact with Patient
Type “BF” (Body Floating): Electrically connected to Patient but not directly to heart
Type “CF” (Cardiac Floating): Electrically connected to the heart of the Patient.
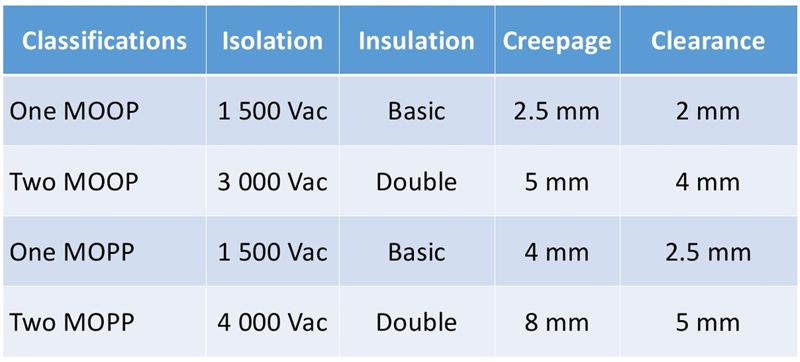
Strengthening patient protection against electrical shock and effects of current leakage, the third revision introduced a more stringent requirement: defining the meaning of different protections for patient and operators. Means of protection (MOP) describes the isolation protection between the electrically charged circuitry and any equipment that may come in contact with the device.
The isolation protection includes the creepage/clearance distances, insulation and protective earth connections. The means of protection is further separated into two categories (Means of operator protection MOOP and Means of patient protection MOPP) implying specific isolation test voltage and creepage distance (see Figure 2).
Click image to enlarge
Figure 02 – IEC 60601-1 third edition, Means of Protection (MOP) classifications in two categories means of operator protection (MOOP) and means of patient protection (MOPP)
The third revision requires from power supply designers to perform a risk assessment analysis in accordance with the ISO 14971:2000 (Application of risk management to medical devices) to ensure that hazards are identified and mitigated to guarantee that the appropriate level of safety provided to the final application.
For example, in a medical equipment not in contact with patient, a power supply complying with 2 x MOOP is normally acceptable though the outcomes from the risk assessment could steer the designer to increase the creepage distance to guarantee higher level of safety for the patients and the operators. IEC60601-1 third revision has introduced a new way of working, combining power design, risk assessment but as well an extreme level of cooperation with equipment manufacturer to guarantee the proper level of safety to patient and operators.
Powering home healthcare safely: IEC 60601-1-11
Collateral standard from the IEC 60601-1, the IEC 60601-1-11 was introduced in 2010 and covers the basic safety and essential performance of medical electrical equipment and systems manufactured for use in the home healthcare environment. Considering the new standard includes a number of major changes compared to previous, manufacturers were given three years to comply. June 30, 2013, the IEC 60601-1-11 went into effect in European markets and by the end of the year in U.S.A and is now empowered internationally.
The latest issue of the IEC 60601-1-11 includes changes in the safety class of medical devices not being installed permanently by an accredited electrician (requirement of Class I products). As the study revealed, and despite significant improvements, EU and US homes have no reliable earth ground wires. In consequence of that, the latest revision requires that all medical devices for home fall under the Class II designation, which does not rely upon earth ground for protection.
In addition, the latest edition addressed the definition of “Nursing Home” by region, which differs between US and Europe. In US nursing home are considered as operated in “Professional” environment (product must meet Class I) when in Europe it’s considered as “Home” environment requiring Class II equipment. This definition is important in case equipment manufactured in USA might not comply with European regulations.
IEC 60601-1-11 also includes additional requirements for interruption of the power supplied to the life-supporting ME equipment or ME systems and to secure the power source (e.g. battery backup) maintains essential performance for a sufficient time or number of procedures, to allow for alternate methods to be employed in such cases.
As collateral to IEC 60601-1 Third revision, the IEC 60601-1-11 requires power designers to consider usability engineering process and file when manufacturing medical products for home use (Clause 9). Not all may apply to power supplies though, when performing the risk assessment it is important to consider all potential impacts. The following hazards must now be considered: Changes of Controls - Confusion in Operation Modes - Unexpected Movement - Transfer of Energy/Substances - Potential of Disconnection - Exposure to Biological Materials - Improper/Unsafe Operation - Parts Inhaled/Swallowed
Powering home healthcare challenges and solutions
Topology and Optimization - taking in consideration standards and regulations, patient comfort and safety, power supply designers are facing a number of challenges, which can all be solved by using proper technologies but as well requiring a significant level of innovation to package always more power in a confined spaced with limited, or no ventilation. Adding to that MTBF and low EMI complying with IEC60601-1-2 and ITE requirements a must, designers’ challenges are becoming very exciting!
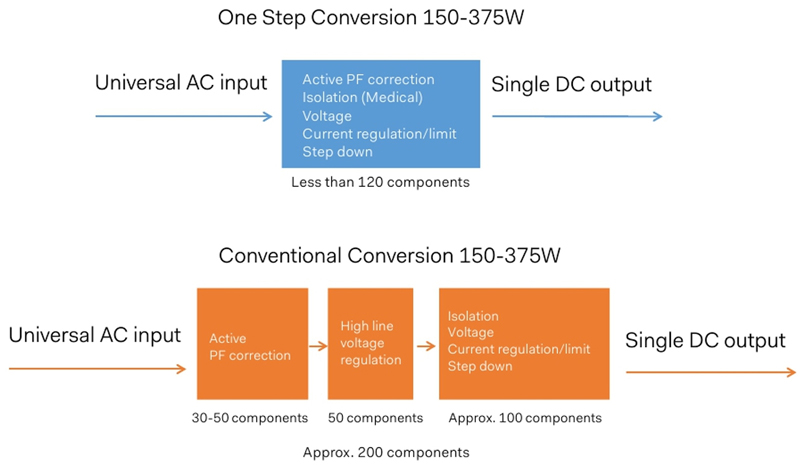
Choosing the right technology at very early stage of the project is a key to success! In the case of external power supplies for medical equipment the topology is an important factor when considering space, cost and performance. Conventional topologies are usually using around to 200 components. Considering an innovative approach, similar to one-step conversion combined with passive power factor correction (PFC) will result in reducing count parts by about 40% with a total of 120 components (see Figure 3) which will directly benefit to the MTBF but also to make the power supply more compact.
Click image to enlarge
Figure 03 – One step conversion technology reduces total components count by 40%
If reducing the number of components to such low number seems to be easy, in practice, it is much more complicated. With a goal to exceed 90% efficiency, a power factor correction >0.90, meeting IEC60601-1-11 requirements, able to operate in free air convection or in sealed box, and to reduce the zero-load power consumption to a level close to 0.3W, power designers have to be very innovative and creative, which can be fun!
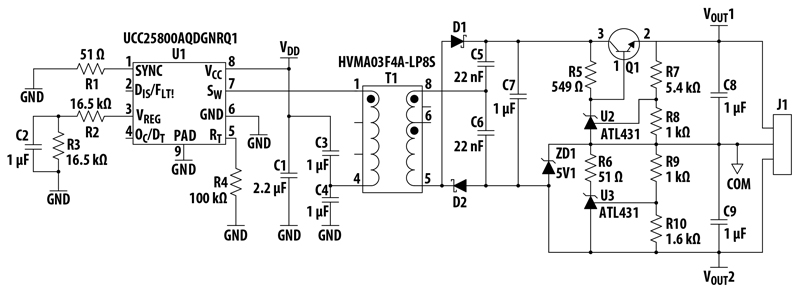
Developing one-step conversion with a limited number of components requires a very high knowledge in transformer and coupling tech to achieve high performance, low leakage current, and EMC compliance with IEC60601-1-2. One example of product based on this approach is showed in Figure 4.
Click image to enlarge
Figure 04 – One step conversion 225W power supply (Powerbox OFM 225) based on One Step Conversion technology
Mechanical with patient in mind
Designing an external power supply for medical applications, especially for home healthcare, requires power designers to work in very close cooperation with end-users to not only secure the highest level of electrical safety, but usability in the patient environment as well.
One example of such cooperation is the development of a new generation of external power supplies for hospital and home healthcare, complying with the IEC60601-1-11 and designed with patient comfort in mind. Conventionally housing for external power supplies for medical equipment over 150W are very similar to PC adapter, rectangular parallelepiped with sharp edges, which in case of homecare might create mobility problems, for example wheelchair blocking. Another parameter often not taken in consideration when designing such type of power supply is when housekeeping cleaning patient’s room. The parallelepiped shapes are not easily cleanable; often at the same time the operators are cleaning the floor, requiring extra attention and time.
Working with a group of users resulted a concept based on a half-lenticular shape, which will avoid wheelchair blocking, make the product easy to clean with conventional Sweepers, and conferring to the product a nice look and feel, which is also very important for patient when such product visible or placed on a desk.
Once the shape was approved by the user group, the next challenge for power designers was to integrate a 225W power supply in the center part of the half-lenticular package and to guarantee that product will operate at full power in patient’s room conditions. With limited number of components and compact design, the one-step conversion makes it possible to contain the power conversion, filtering, and monitoring in the limited space and operate safely without the need for extra cooling (see Figure 5).
Click image to enlarge
Figure 05 – Home Healthcare external power supply (Powerbox EXM 225) designed in semi lenticular shape for homecare integration and patient comfort
Another important safety aspect is to guarantee that input and output cables are secured by proper locks to avoid power supply disruption in case cables are pooled out accidentally. AC input connector must be protected and locked to sustain a traction of 100 Nm pull strength while the output connector secured by mechanical lock, requiring an action from the operator to unlock (see Figure 6).
Click image to enlarge
Figure 06 – Output connector equipped with lock to prevent accidental disconnection and power disruption to medical equipment
Looking forward
Designing a power supply for medical application, operated in home healthcare environment, requires power designers to fully understand and comprehend how the final product will be operated. Taking in consideration end users, in this specific case the patient environment, is business critical.
References:
Hospital Noise and Patients’ Wellbeing
www.sciencedirect.com/science/article/pii/S1877042815001470
IEC60601-1-11
www.iso.org/iso/home/store/catalogue_ics/catalogue_detail_ics.htm?csnumber=65529