Recent Progress of Carbon-Based Non-Noble Metal Single-Atom Catalysts for Energy Conversion Electrocatalysis
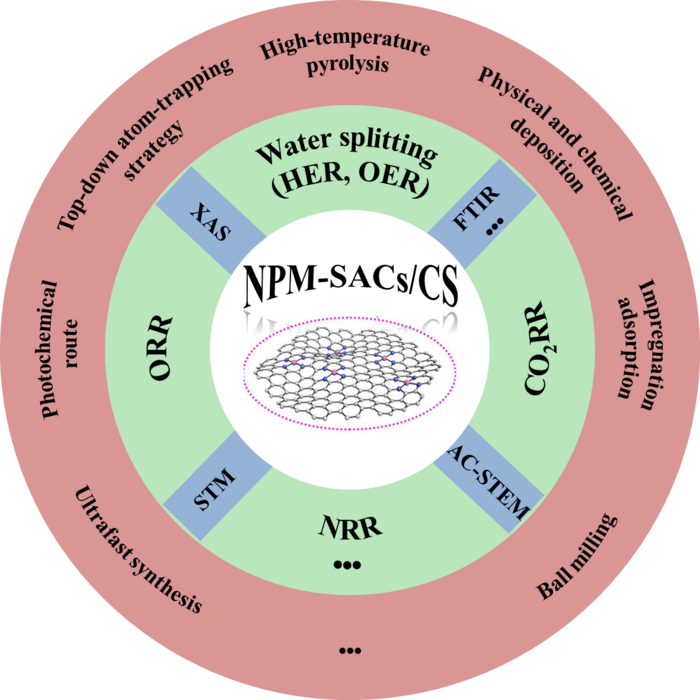
This review is led by Prof. Yuehe Lin (Washington State University), Prof. Welei Zhu (Nanjing University), and Prof. Yuanyuan Wang (Nanjing University), they provided an overview of recent developments in non-precious metal single-atom catalysts (NPM-SACs) for the electrocatalytic field. NPM-SACs with unique electronic structures and coordination environments has gained much attention in electrocatalysis owing to their low cost, high atomic utilization, and high performance. NPM-SACs on carbon support (NPM-SACs/CS) are promising because of the carbon substrate with a large surface area, excellent electrical conductivity, and high chemical stability.
First, the team not only introduced some widely used methods, such as high-temperature pyrolysis, physical and chemical deposition, impregnation adsorption and etc, but also collected some other innovative and unique ultrafast synthesis method reported recently. For characterization, the team classified scanning tunneling microscopy (STM) and aberration-corrected high-angle annular dark-field scanning transmission (AC HAADF-STEM) for morphological observation, X-ray absorption spectroscopy (XAS) for atomic structure and electronic property studies, and other specific characterizations for auxiliary analysis.
Then, the team summarizes strategies to optimize the macro- and micro-environment of NPM-SACs/CS. The microenvironment here mainly includes adjusting the type of central metal atom, changing the coordination number of a nitrogen atom, building non-nitrogen heteroatom coordination, adjusting carbon basal plane, and building bimetallic sites.
Furthermore, the team some comprehensive summary of recent advances in developing NPM-SACs/CS for important electrochemical reactions, including carbon dioxide reduction reaction (CO2RR), hydrogen evolution reaction (HER), oxygen evolution reaction (OER), oxygen reduction reaction (ORR), and nitrogen reduction reaction (NRR).
Last, the team highlighted the existing challenges and future opportunities of NPM-SACs/CS in the electrocatalytic field, involving the control synthesis of high metal loading NPM-SACs/CS, the development of advanced single-atom characterization techniques, the exploration of structure-activity relationship of NPM-SACs/CS for electrochemical reactions, construction of new coordination environments for boosting the catalytic performances, and the improvement of catalytic stability under reaction conditions.
EurekAlert!, the online, global news service operated by AAAS, the science society.


.jpeg)


