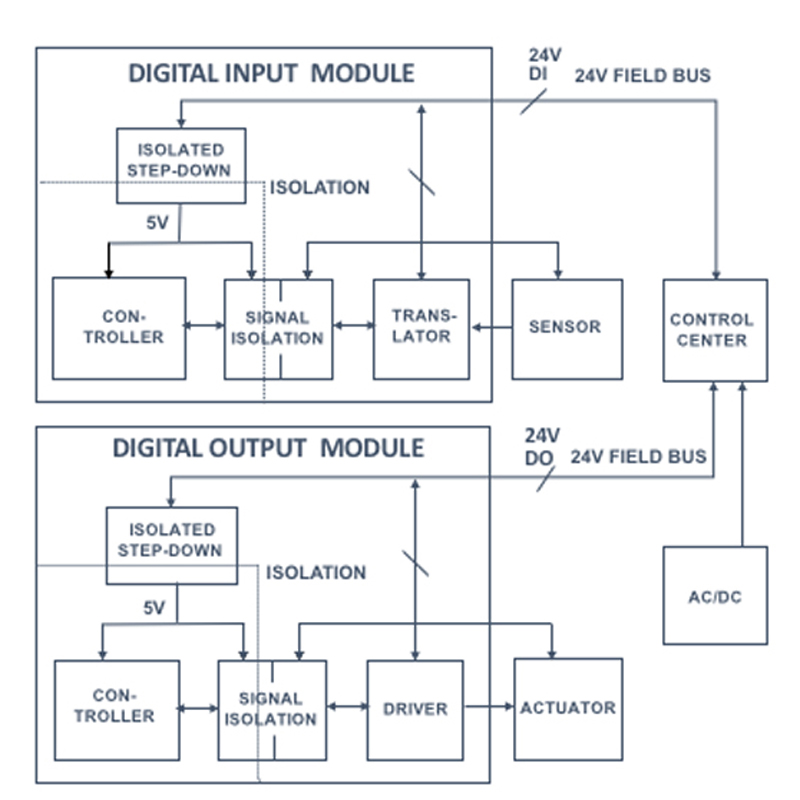
Researchers Boost Lifetime of Sodium-Ion Batteries

In the race to bring new battery chemistries to market, sodium-ion batteries possibly holds the best potential. However, there is one obstacle that needs to be overcome to commercialize the technology. Sodium-ion, in fact all sodium based batteries, have a problem with longevity. They simply lose the ability to hold a decent amount of charge over a relatively short amount of charge/discharge cycles. Sodium based batteries have some strong capabilities in that they are more resistant to temperature change and they are very stable. They may not be as energy dense as lithium based batteries so far, but if the lifecycle problem could be fixed, they could offer a cheap alternative to lithium, especially for applications like grid energy storage.
A research team from the Department of Energy’s Pacific Northwest National Laboratory (PNNL) has been working to improve sodium-ion batteries and has now produced a model that has extended the longevity of the technology in laboratory tests. The team has changed the ingredients that make up the liquid core of the battery to stop the performance issues that have prevented sodium-based batteries fulfilling their potential.
In batteries, the electrolyte forms by dissolving salts in solvents, resulting in charged ions that flow between the positive and negative electrodes. Over time, the electrochemical reactions that keep the energy flowing get sluggish, and the battery can no longer recharge. In current sodium-ion battery technologies, this process happens much faster than in similar lithium-ion batteries. The new sodium-ion battery technology keeps that ability for longer than previous types of sodium-ion batteries.
The PNNL team, led by scientists Yan Jin and Phung Le, switched out both the liquid solution and the salt flowing through it to create a brand new electrolyte recipe. In laboratory tests, the new design held 90 percent of its cell capacity after 300 cycles at 4.2 V. The current electrolyte recipe for sodium-ion batteries results in the protective film on the anode dissolving over time. This film allows sodium ions to pass through while preserving battery life. The new recipe stabilizes this protective film. The electrolyte also generates an ultra-thin protective layer on the cathode that contributes to additional stability of the entire unit.
The new sodium-ion technology also uses a naturally fire-extinguishing solution that is impervious to temperature changes and can operate at high voltages. This feature is helped by the ultra-thin protective layer that forms on the anode, which remains stable once formed, providing a longer cycle life.
“We also measured the production of gas vapor at the cathode,” said Phung Le, a PNNL battery chemist and one of the lead authors of the study. “We found very minimal gas production. This provides new insights to develop stable electrolyte for sodium-ion batteries that may operate at elevated temperatures.”
The study was supported by the Department of Energy’s Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office. The findings of the study have been published in the journal Nature Energy. The research team will continue to refine its design, including to reduce, and eventually eliminate, cobalt, which is toxic and expensive if not recovered or recycled.
.jpg)