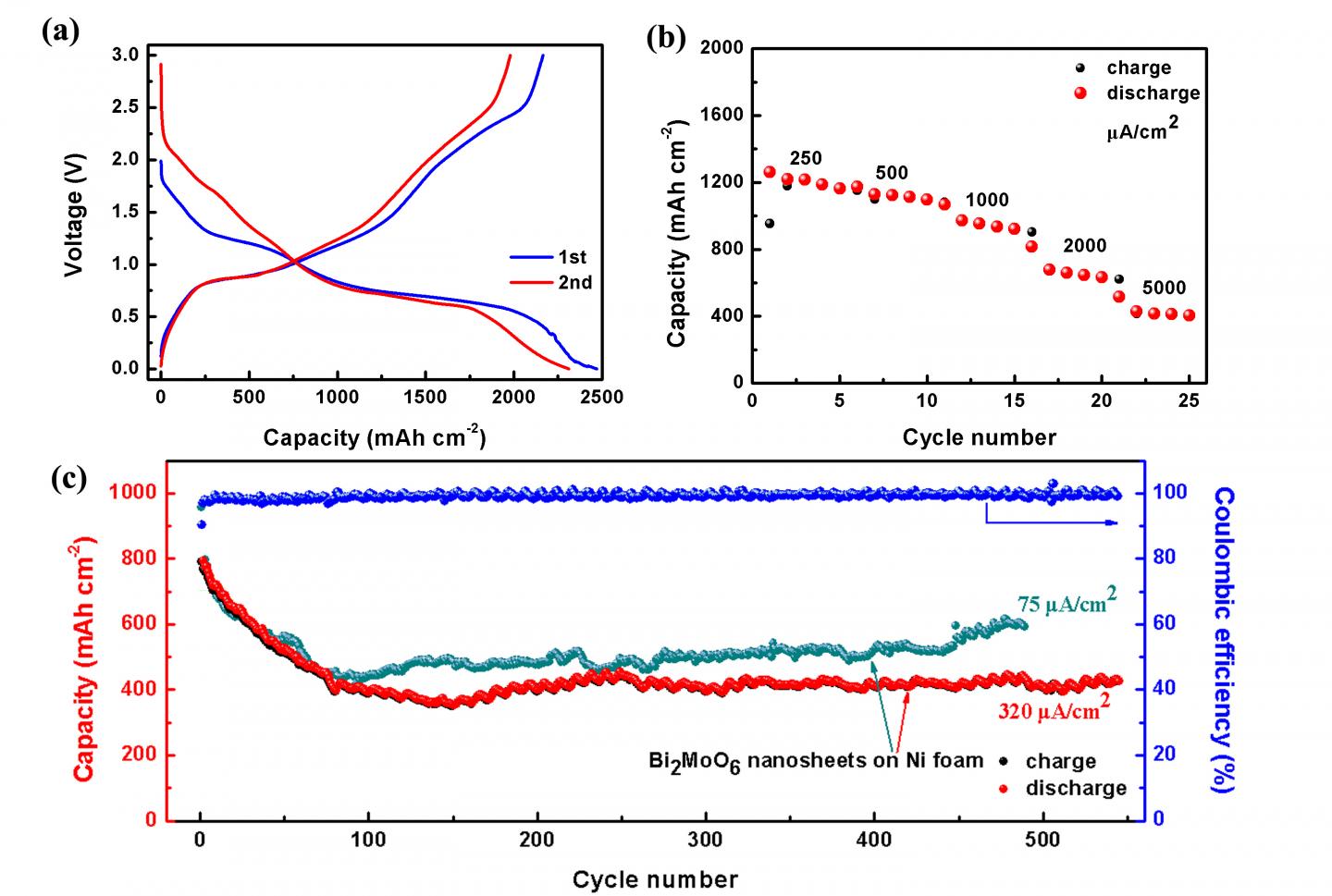
Recent research published in a paper in NANO by a group of researchers from Northeastern University investigate the effect of hierarchical Bi2MoO6 nanosheet arrays growing on three-dimensional Ni foam synthesized by one-step template-free route. The obtained BNAs used directly as binder-free integrated electrode for Li-ion batteries (LIBs) exhibits a super high reversible discharge capacity of 2311.7 μAh/cm2, and an excellent cycle stability.
With the rapid development of modern technology, a variety of portable electronic products have become the requirement of time. Li-ion batteries (LIBs) are the optimum selection by virtue of long cycle-life and high energy density properties. Meanwhile, LIBs are viewed as one of the most promising technology in various fields including the defense industry, space technology, electric vehicles and other fields. Nowadays, commercial LIBs mainly use graphite as the anode material. However, graphite can hardly provide the high capacity and high energy density necessary to satisfy the demand required for high power application of the next-generation LIBs due to its low theoretical specific capacity.
Thus, development of high-performance anode materials with high capacity as well as low insertion voltage is urgently needed. In terms of their high capacity, lots of metal oxides have attracted great interest in recent years. However, most metal oxides have low conductivity, high desertion voltage and structural instability, which result in poor rate capability, low power density and poor cycling stability. These shortcomings limit the application of metal oxides as anode materials for LIBs.
Among the numerous metal oxides, Bi2MoO6 with high theoretical capacity (791 mAh/g) and low desertion voltage (<1.0 V) has been widely studied for its excellent photoelectric properties, but, there are few reports on the nanostructure Bi2MoO6 as anode material for LIBs, not to mention the Bi2MoO6 integrated electrode.
Thereafter, developing an effective strategy to prepare the Bi2MoO6 integrated electrode and then exploring their electrochemical performance toward lithium is of great importance. To tackle the issue of low intrinsic conductivity, inferior cycling stability for Bi2MoO6 as anode material, the researchers proclaim an effective strategy to the synthesis of hierarchical Bi2MoO6 nanosheet arrays (BNAs) on the three-dimensional (3D) Ni foam by one-step template-free hydrothermal method. Remarkably, BNAs-integrated electrodes exhibit excellent electrochemical properties (a super high reversible discharge capacity of 2311.7 μAh/cm2, more than 500 times of cyclic stability), when used as the anode electrode for LIBs.
Undoubtedly, this work disclosed a new comprehension for improving the performance of LIBs with metal oxides as the anode material. It can improve the cycle stability and capacity of lithium-ion battery and is expected to be used in portable electronic devices in the future.
EurekAlert!, the online, global news service operated by AAAS, the science society: https://www.eurekalert.org/pub_releases/2018-06/ws-bip062718.php