Lithium-sulfur technology comes closer to challenging Li-ion
Argonne National Laboratory researchers find adding a layer to Li-S batteries provides both energy storage capacity and eliminates shuttling problem
Batteries are the hottest topic in the technology field. Many chemistries are being investigated to try either replace the Li-ion batteries we use today, or to provide a solution for an individual application and save on the amount of lithium that is extracted. Although none of the research has really come to fruition so far, scientists are getting closer and closer all the time. Li-ion batteries have worked well for us and empowered a wave of innovation, but they have major problems themselves, and other battery chemistries may prove safer and more efficient. For example, Li-ion batteries have short lifetimes, overheat too easily and the raw materials that they required for their construction are difficult to source.
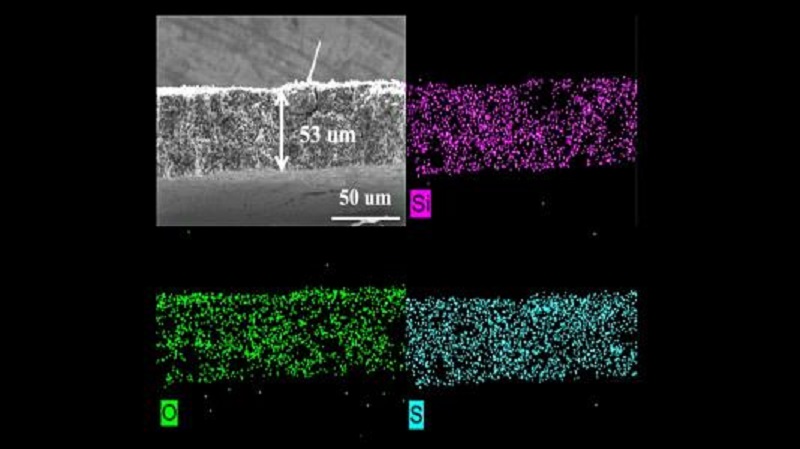
Lithium-sulfur (Li-S) batteries should overcome the majority of these problems, and the chemistry is a favourite to replace Li-ion batteries if certain obstacles can be removed. Sulfur is a very common element and the Li-S batteries have the potential to store more energy than Li-ion ones. However, the first drawback is that in a lithium-sulfur battery, polysulfides dissolve into the electrolyte, causing corrosion. This polysulfide shuttling effect negatively impacts battery life and lowers the number of times the battery can be recharged. One solution to polysulfide shuttling is adding a redox-inactive interlayer between the cathode and anode, meaning that the material does not undergo reactions like those in an electrode. However, the protective interlayer required turned out to be heavy and dense, which reduced the energy storage capacity per unit weight for the battery. The barrier also did not reduce shuttling as much as expected.
Now, researchers have built an advanced sulfur-based battery by using a porous sulfur-containing interlayer. Tests in the laboratory with the interlayer showed that the initial capacity was about triple that of Li-S cells with an inactive interlayer. The cells with the active interlayer also maintained high capacity over 700 charge-discharge cycles.
“Previous experiments with cells having the interlayer suppressed the shuttling, but also sacrificed the energy for a given cell weight,” said Guiliang Xu, an Argonne chemist and co-author of the paper. “By contrast, our redox-active layer adds to energy storage capacity and suppresses the shuttle effect.”
The research team conducted experiments at the 17-BM beamline of Argonne’s Advanced Photon Source (APS). The data gathered from exposing cells with this layer to X-ray beams confirmed that a redox-active interlayer can reduce shuttling, reduce detrimental reactions within the battery and increase the battery’s capacity to hold more charge, as well as lasting for more cycles. With this drawback removed, Li-S batteries come one step closer to commercialization.
A paper based on the research appeared in Nature Communications. Khalil Amine, Tianyi Li, Xiang Liu, Guiliang Xu, Wenqian Xu, Chen Zhao and Xiao-Bing Zuo contributed to the paper.
The Argonne National Laboratory is part of the U.S. Department of Energy (DOE). This research was sponsored by the DOE’s Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office Battery Materials Research Program and the National Research Foundation of Korea.





