How to minimise the risk of performance drops or loss of functionality in medical devices
Although essential performance is covered in IEC 60601-1 2nd edition, the 3rd edition defines it in greater detail as “the performance necessary to achieve freedom from unacceptable risk”. It is up to the manufacturer to determine if a loss of performance or functionality will result in an acceptable risk or an unacceptable risk. This is analysed by determining the probability or the frequency of an event happening and the severity of that event. If a medical device loses performance or functionality in a way that it harms a patient, the operator or the environment, that is classed as an unacceptable risk.
As an example, if a handheld battery-operated meter that measures blood glucose levels was to stop working due to a defective part, it would be an acceptable risk. This is because they are usually readily available in a hospital or clinic, and a replacement could be easily found. If a failure caused the device to display an incorrect reading, it could result in harm to a diabetic patient if too much or too little insulin was then administered. That failure would certainly be an unacceptable risk.
Although medically certified power supplies are not classed as medical devices, their (EMC immunity) performance under IEC 60601-1-2 can affect the medical device it is powering, particularly with the stricter IEC 60601-1-2:2015 4th edition levels now published.
IEC 60601-1-2 is based on the IEC 61000-4 standard, and for input voltage dips IEC 61000-4-11 applies. This standard covers “Testing and measurement techniques - Voltage dips, short interruptions and voltage variations immunity tests”.
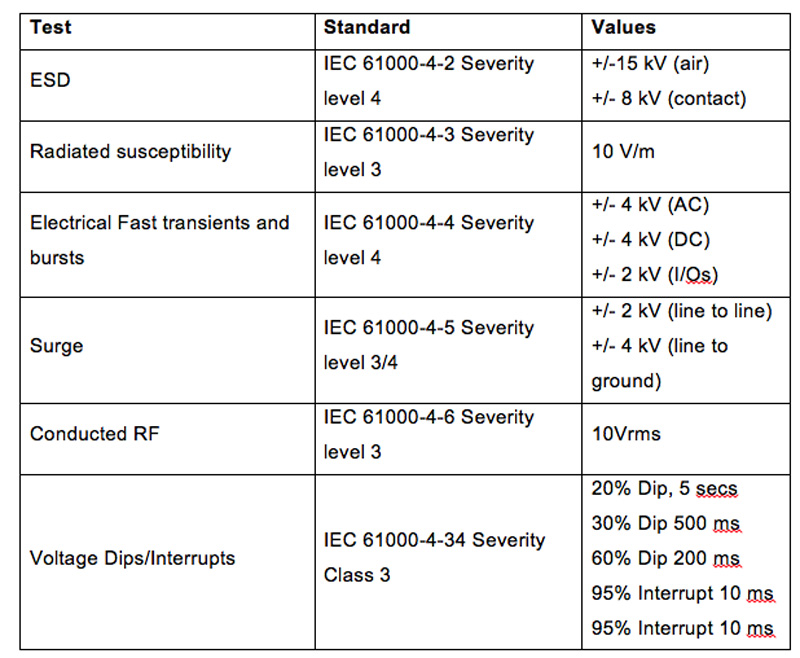
There are five key tests indicated in Table 1. 100Vac input and 50Hz conditions are shown as they could represent the worst case.
Click image to enlarge
Table 1: IEC 61000-4 standard, and for input voltage dips IEC 61000-4-11
Tests are made against four performance criteria levels:
Performance Criteria A – ‘Performance within specification limits’
This is the most acceptable result. A small voltage transient on the output may be similar to that seen for a step load change and is unlikely to cause the end equipment to malfunction.
Performance Criteria B – ‘Temporary degradation which is self-recoverable’
Criteria B would probably be acceptable in most cases. This could occur during a short interruption to the AC input.
Performance Criteria C – ‘Temporary degradation which requires operator intervention’
This performance criterion would generally be classified as unacceptable from a user point of view, without even considering a risk analysis. If the AC power dropped out for a few cycles and the power supply had to be reset by a nurse or doctor, it would be too inconvenient.
Performance Criteria D – ‘Loss of function which is not recoverable’
Criteria D is a “fail” recording on the test result in that the power supply had suffered damage and no longer functions. It is highly unlikely that any manufacturer would place a product with this performance level on the market.
Referring to Table 1, most power supplies will pass the first two tests with a Performance Criteria level A with some output derating to increase the hold-up time.
The third and fourth tests require the power supply to continue to operate for 200ms when the input drops to 40% of nominal or for 500ms at 70% of nominal. This would be 40Vac with an AC supply voltage of 100V. There is a way to still achieve Criteria A though. Modifying the product’s low voltage input protection circuitry would allow the power supply to operate at the low input voltage for a short time. As the AC input current will be higher, the user must ensure that the power supply is not operated at full load, which many users already do to improve product life. As hold-up is related to the actual output power drawn, operating the power supply at 50% load will result in a significant increase in the hold-up time.
All of the above tests are of course much easier to comply with if the medical device is operated with a 208/230Vac input.
The last test of a 5 second interruption to the AC supply is usually met by the medical device manufacturer, with the installation of battery back-up or a UPS (Uninterruptable Power Supply). Adding sufficiently large energy storage electrolytic capacitors inside the power supply would result in a significant increase in size.
As mentioned, the risk analysis has to be conducted by the medical device manufacturer. Accepting Criteria below level A will depend always upon the end application, and if harm will occur.